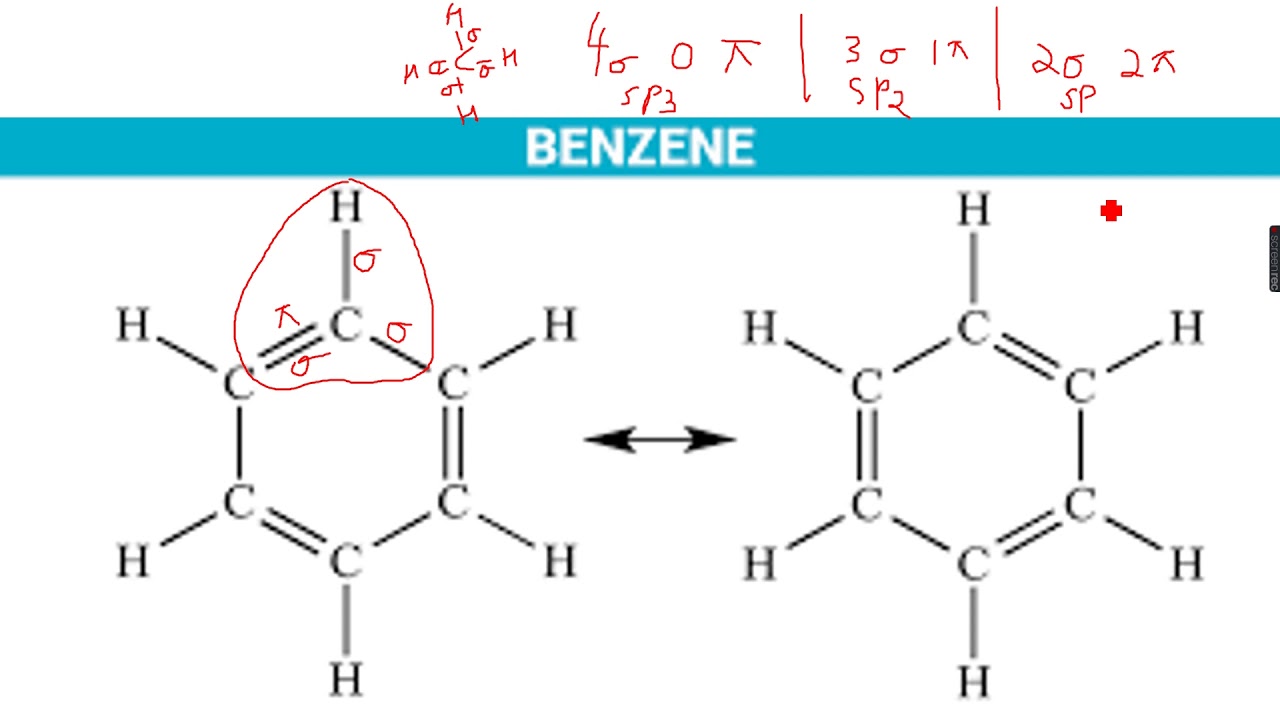
A dashed line indicates the solitary link between the carbon and oxygen atoms. The carbon atom is connected to a single oxygen atom and a hydrogen atom in the third resonance structure. The carbon atom is connected to a single oxygen atom and a second carbon atom in the second resonance structure. A dashed line indicates the double bond between the carbon and oxygen atoms. The carbon atom is connected to two oxygen atoms in the first resonance structure. Numerous contributing structures are used to depict a molecule or ion with such delocalized electrons (also called resonance structures or canonical forms). Resonance is a term used to describe delocalized electrons within specific compounds or polyatomic ions whose bonding cannot be represented using a single Lewis formula. The electrons in a resonance structure participate in more than one covalent bond, and the electron pairs are shared between the atoms in diverse ways.Įven when formal charges are taken into account, the bonding of certain molecules or ions cannot always be described by a single Lewis structure. The resonance structures are drawn with the same link lengths and angles, and the electrons are dispersed in the same way between the atoms. The different structures are called resonance structures because they “resonate” with each other, implying that they are all equally acceptable representations of the molecule. Resonance structures are capable of explaining delocalized electrons that cannot be described in an integer number of covalent bonds using a single Lewis formula. The term “resonance structure” refers to a collection of two or more Lewis Structures that together describe the electronic bonding of a single polyatomic species, including fractional bonds and charges.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
